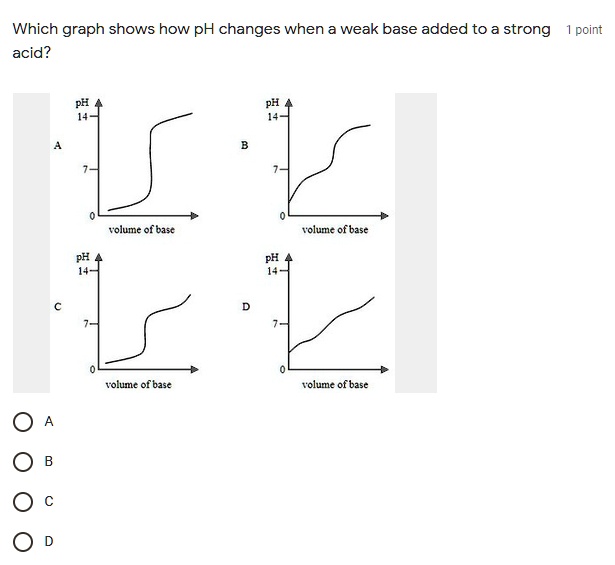
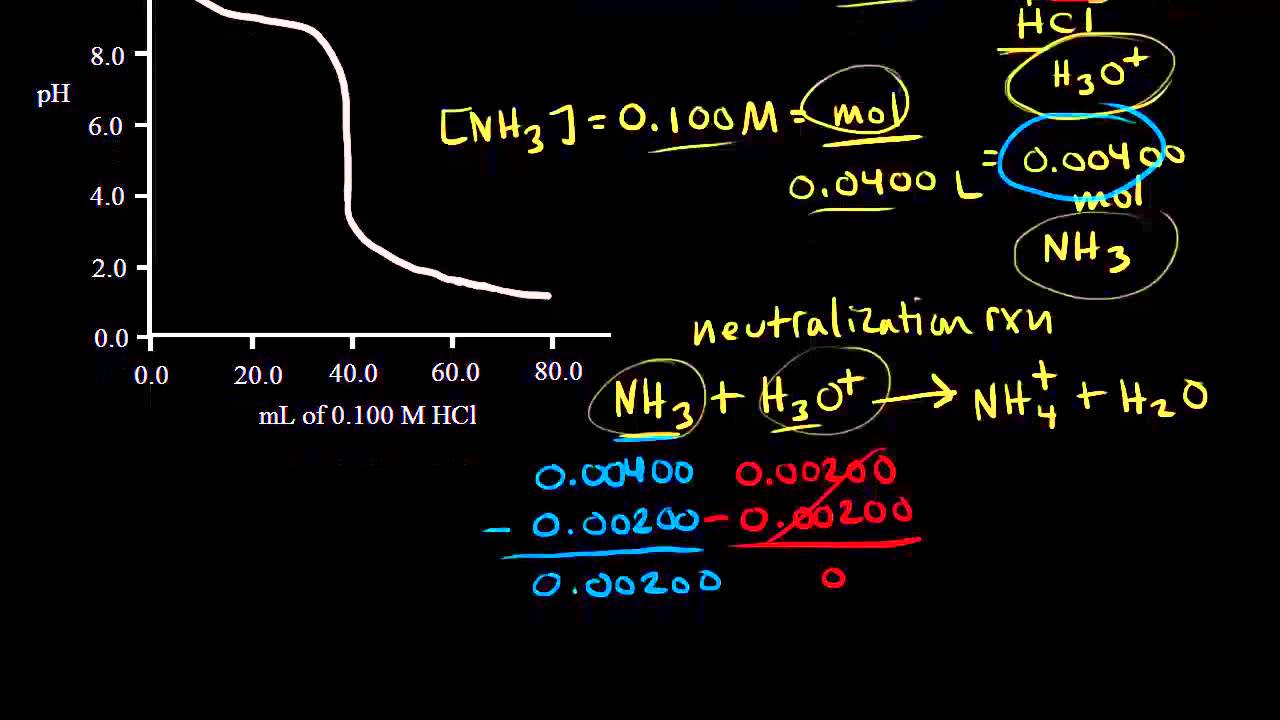
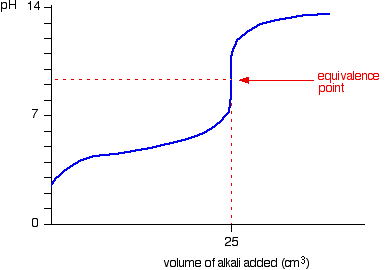
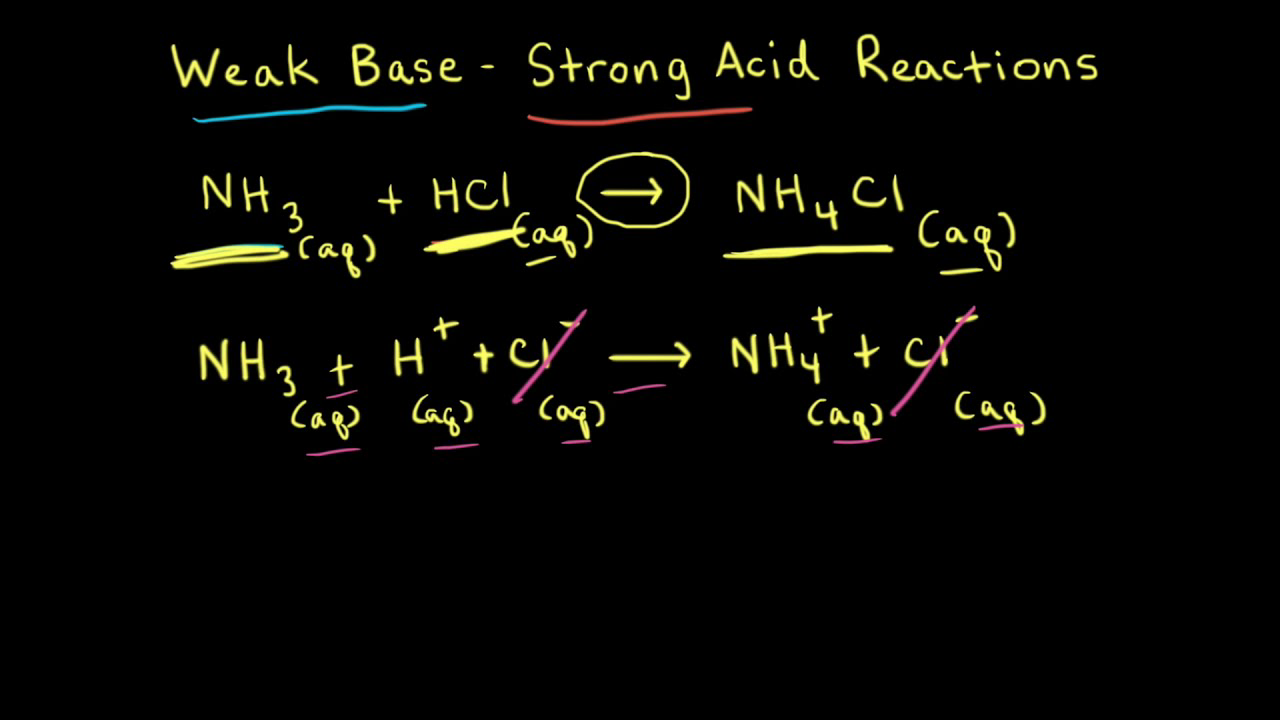
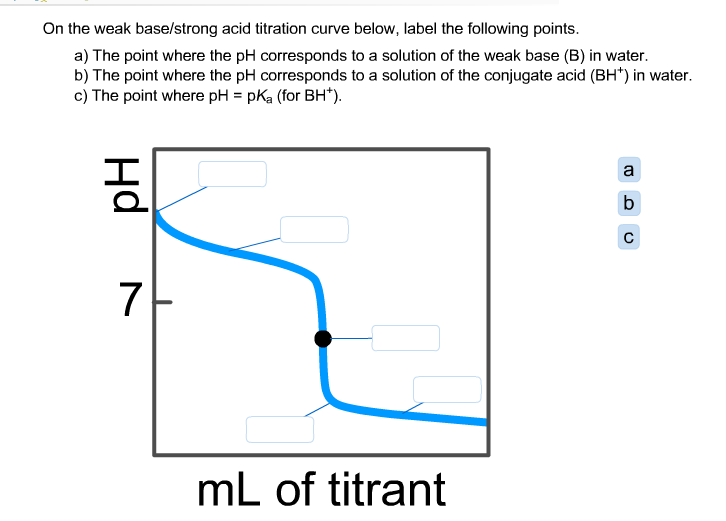
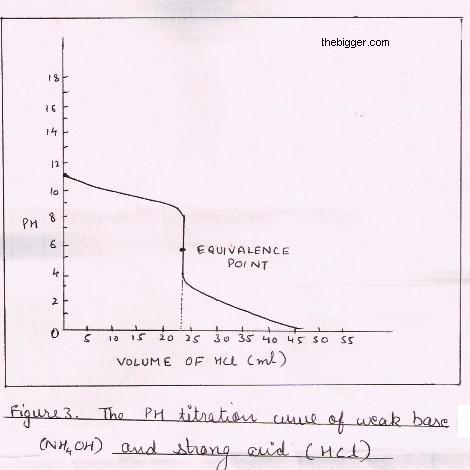
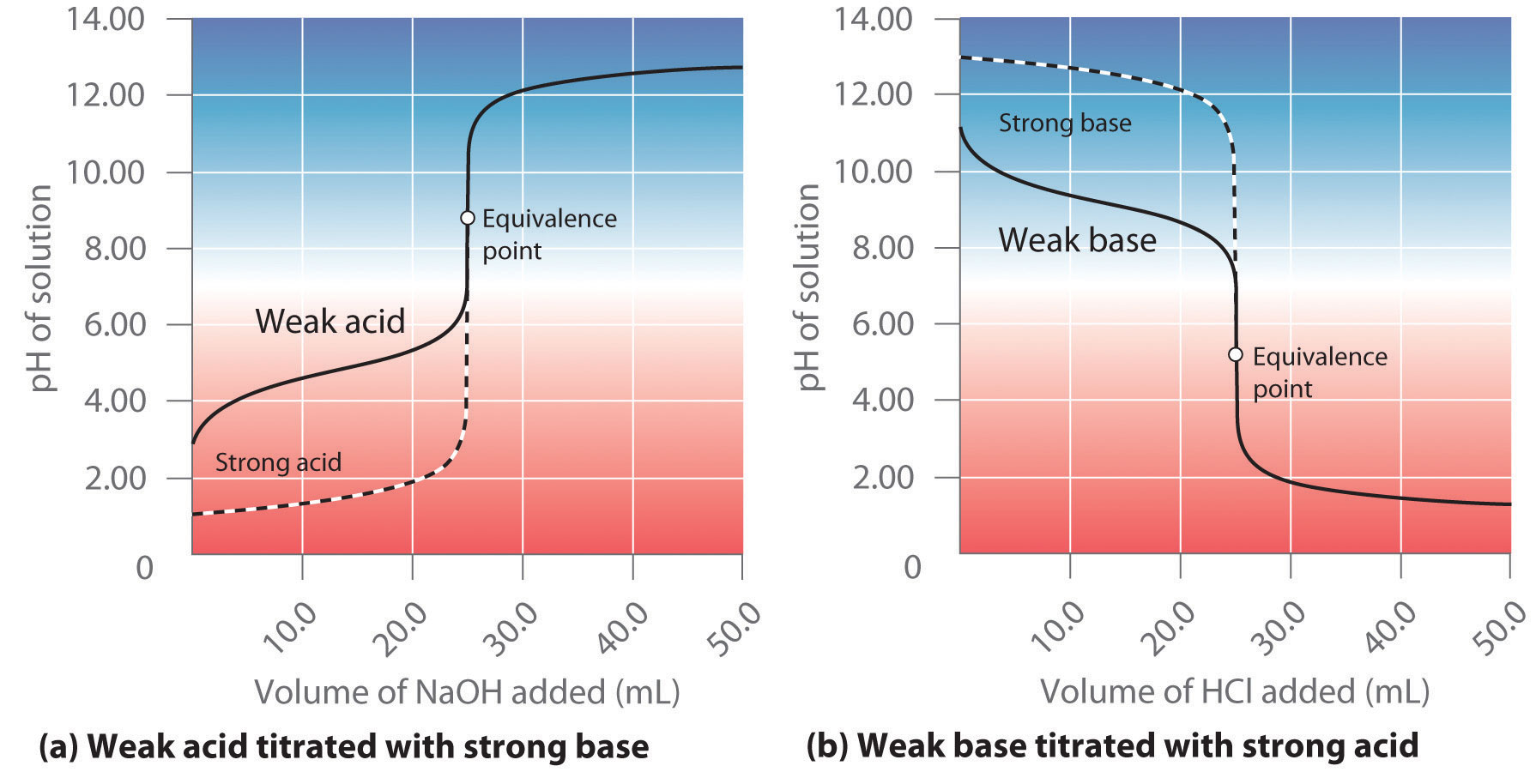
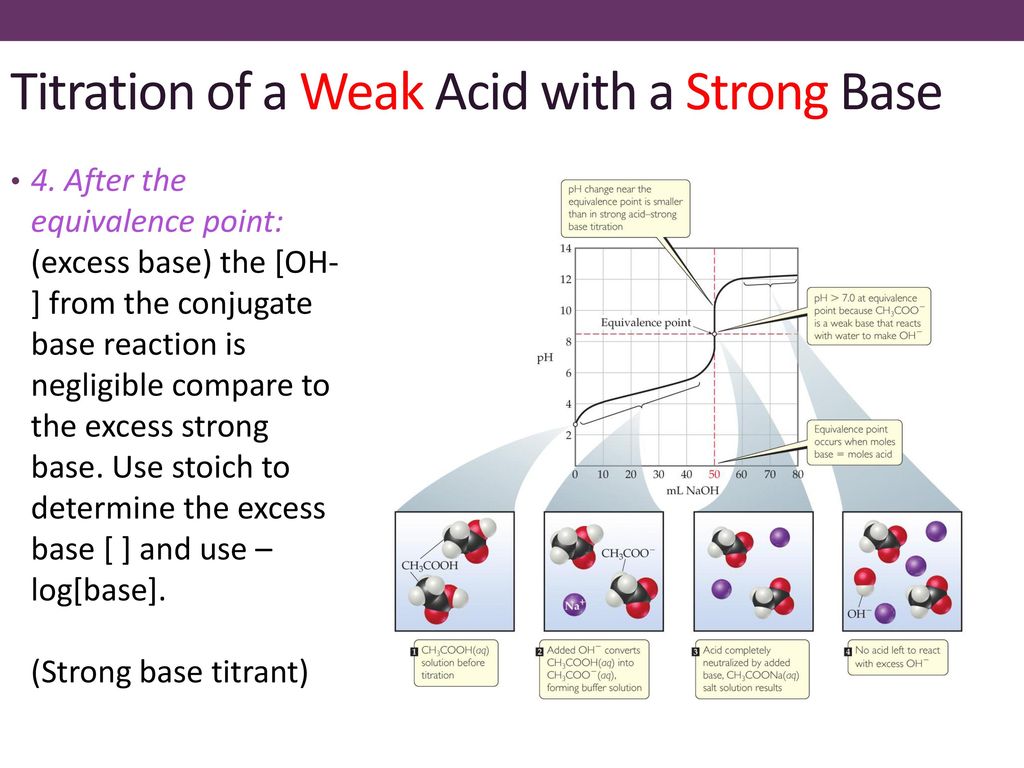
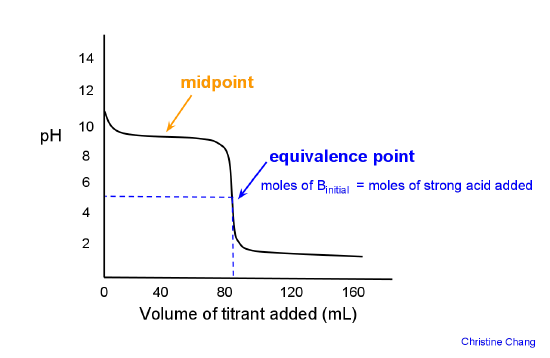
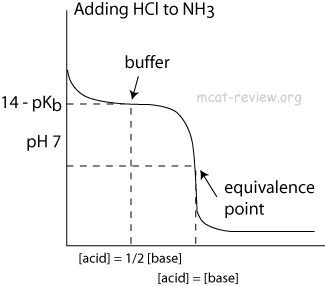
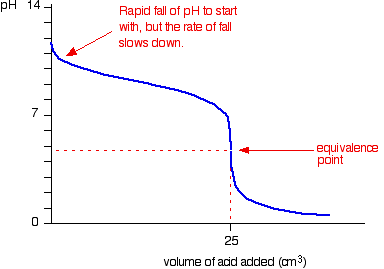
Sketch the following titration curves. a) A strong acid/strong base. b) A weak monoprotic acid/strong base. c) A weak diprotic acid/strong base. | Homework.Study.com
Which of the following is correct?(A) K (weak acid). K (conjugate weak base)= Kw (B) K (strong acid). K(conjugate weak base)= Kw (C) K( weak acid). K(weak base)=Kw (D) K (weak acid).