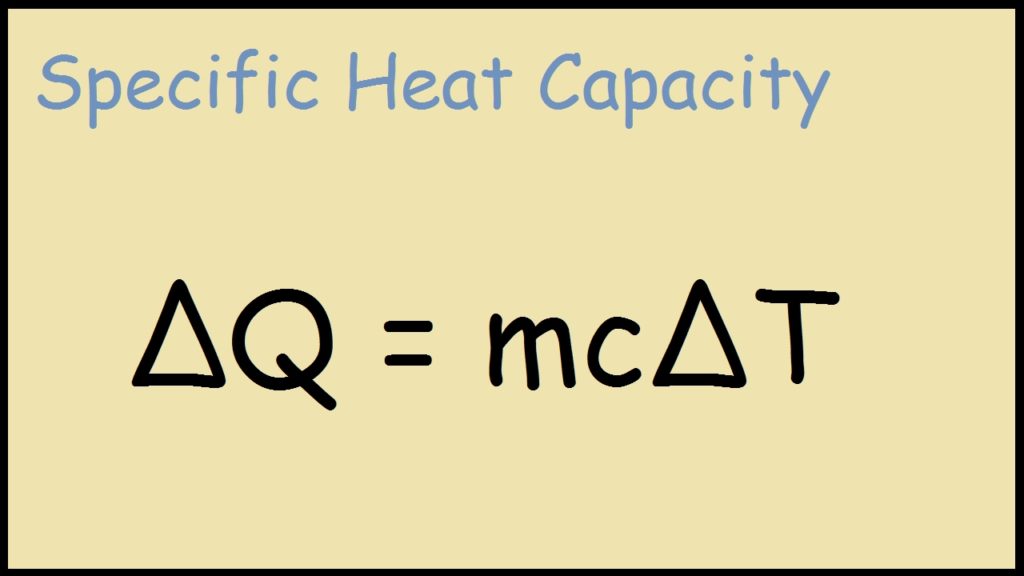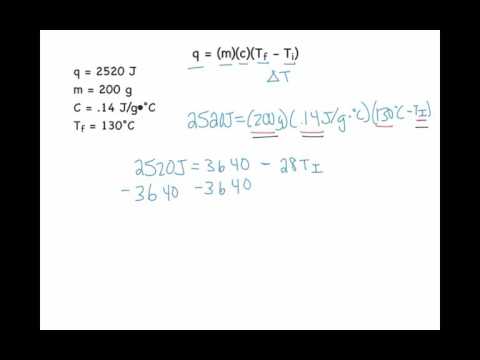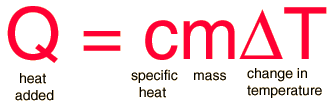SOLVED: Consider the following problem; A sample of copper absorbs 43 kJ of heat; resulting in a temperature rise of 75.0'C, Determine the mass of the copper sample if the specific heat

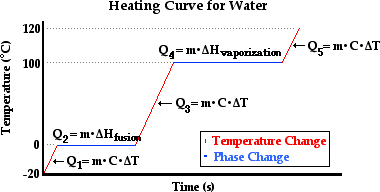
Chapter 10: Energy Problems (ME fusion/ vaporization) and Q=mc delta t | Science, Chemistry | ShowMe