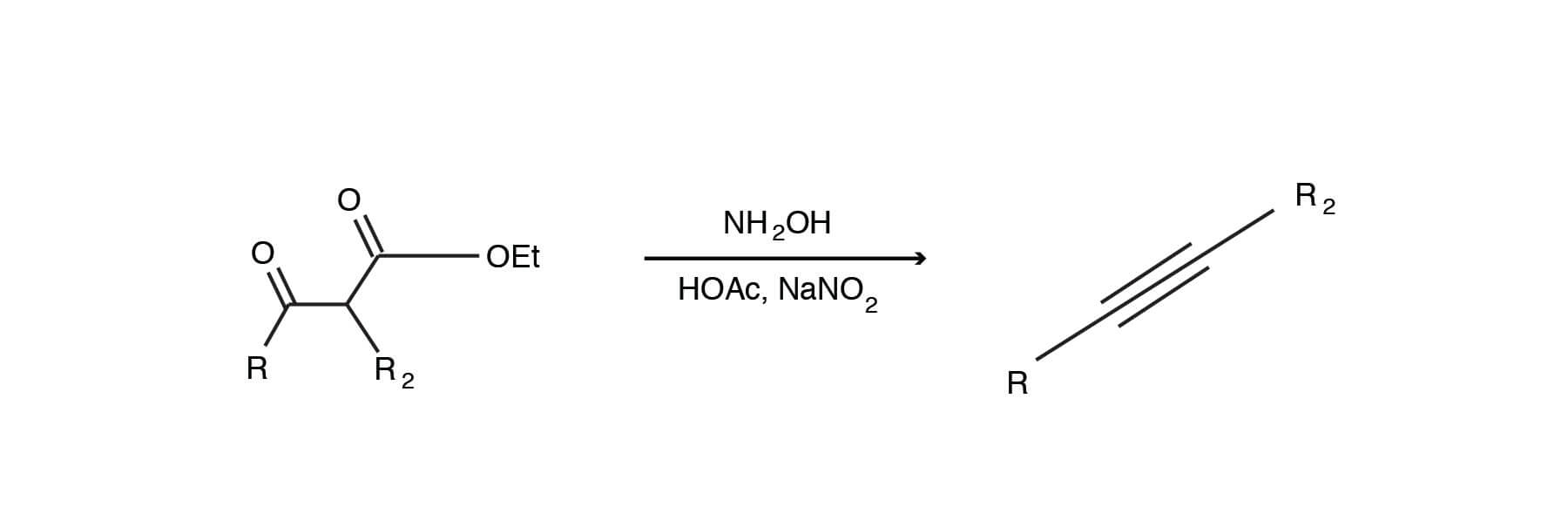
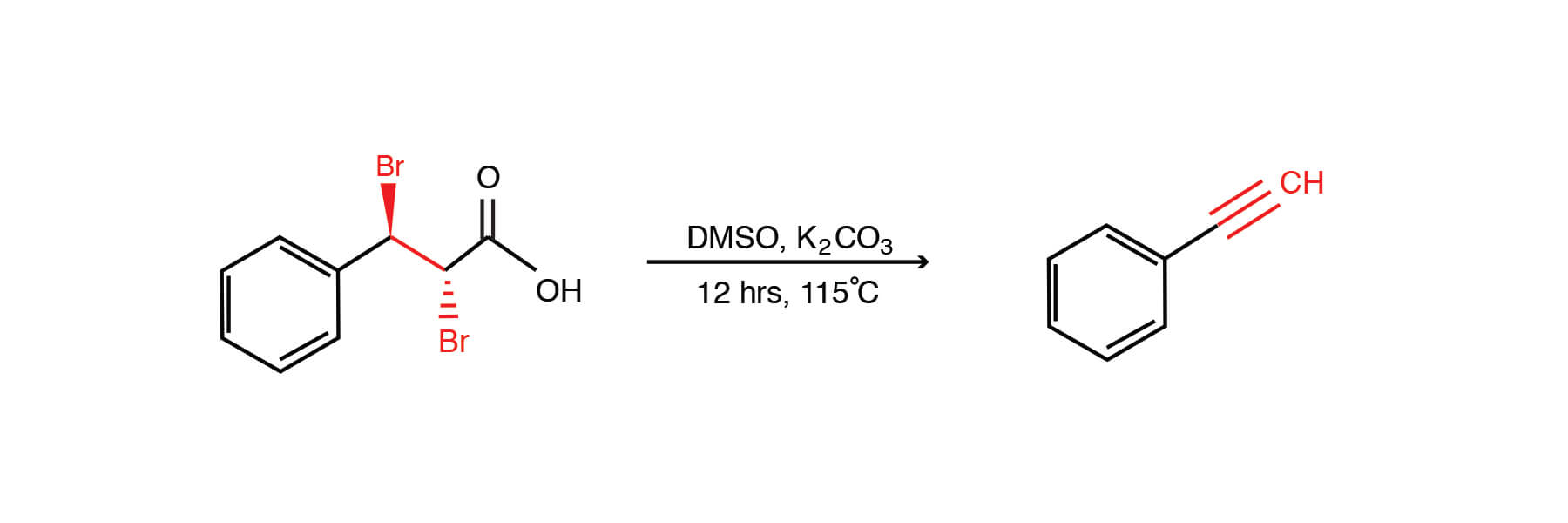
Catalytic Hydroboration of Aldehydes, Ketones, and Alkenes Using Potassium Carbonate: A Small Key to Big Transformation | ACS Omega

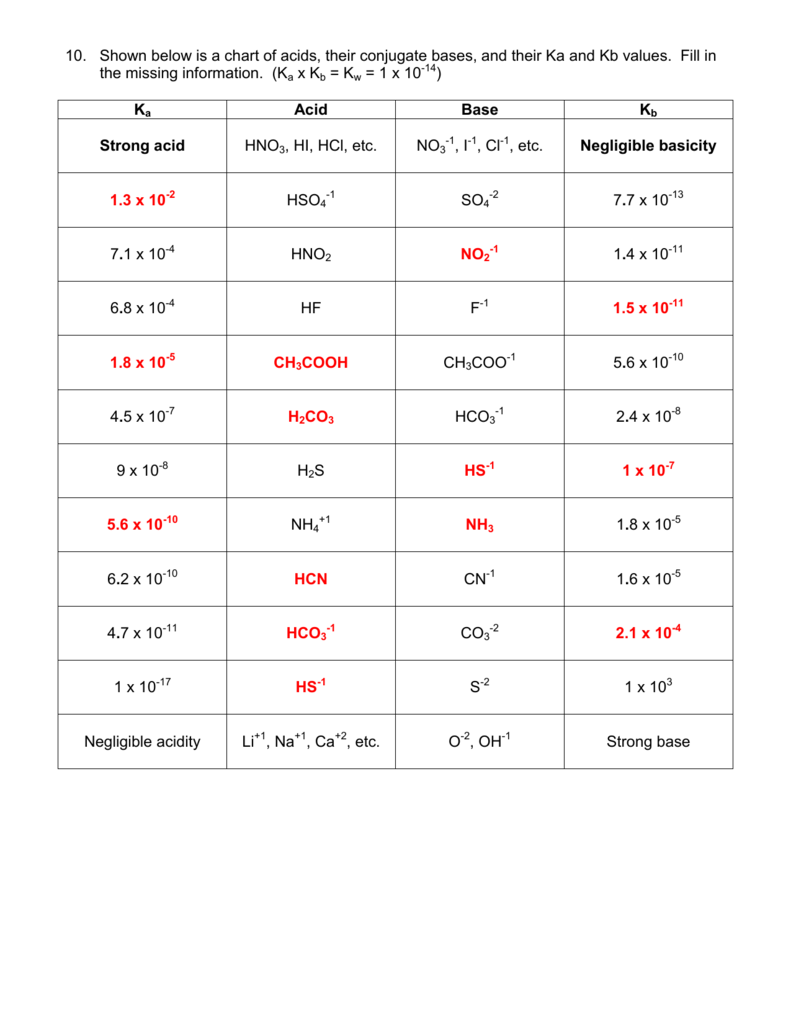
Solid Acid/Base Catalysis in Sub- and Supercritical Water | Industrial & Engineering Chemistry Research
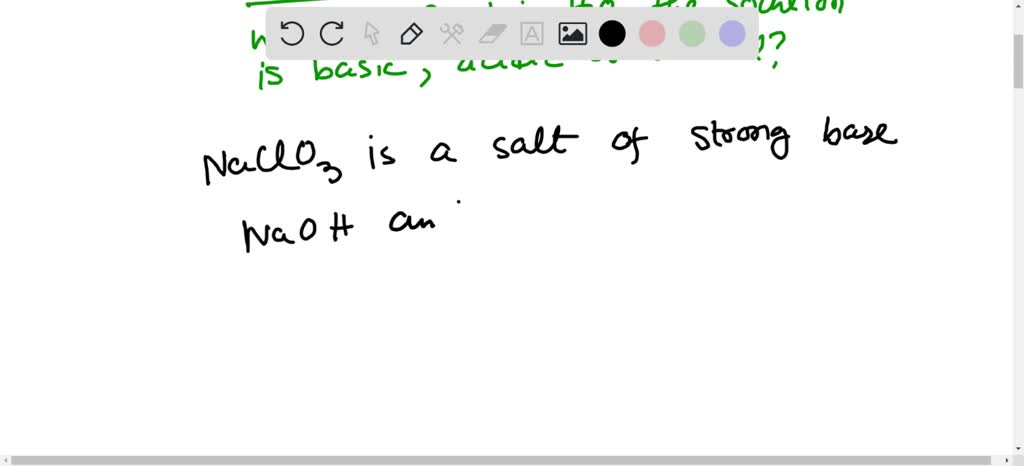
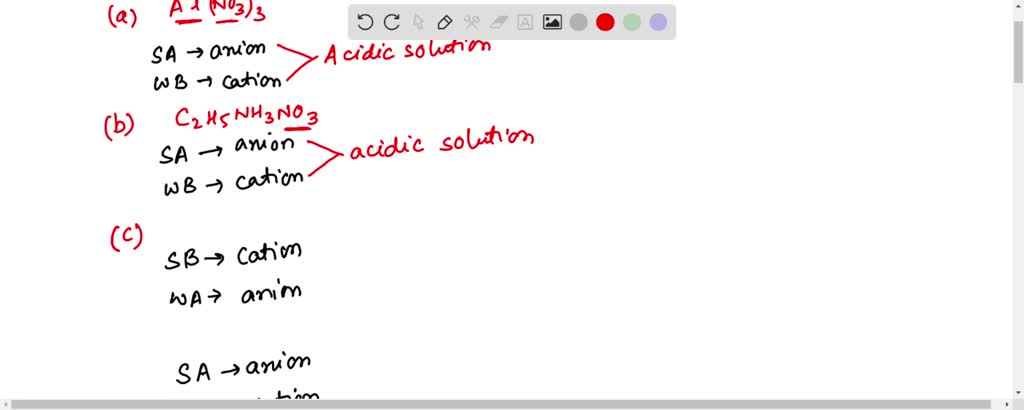
SOLVED: When K2CO3 is dissolved in water, will the solution be acidic, basic, neutral, or will more information be needed to decide?
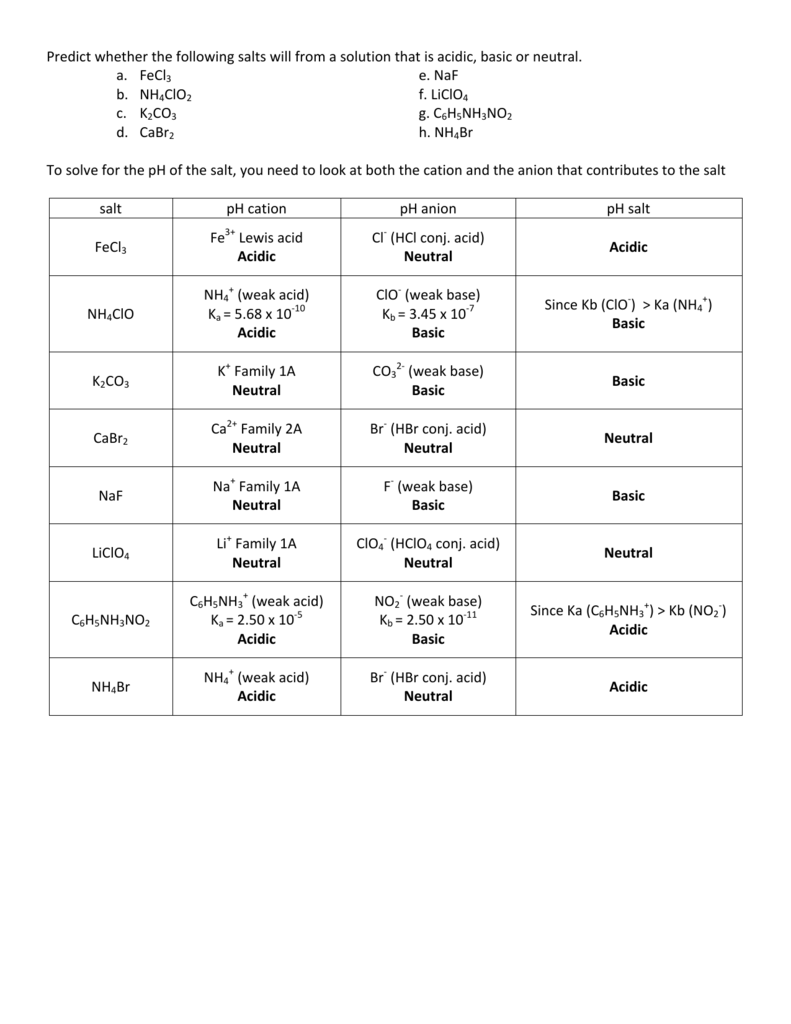
How would you determine is the following salts will from a solution that is acidic, basic, or pH neutral? CH3NH3CN, Fe(ClO4)3, K2CO3, CH3NH3CL, RbI | Socratic

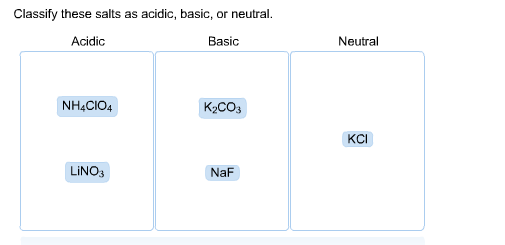
Classify these salts as acidic, basic, or neutral. And Why? NH4ClO4 , KCl , LiNO3 , NaCN , K2CO3 - YouTube

SOLVED: Select the salt from the list below which will produce a basic aqueous solution. Group of answer choices NaNO3 K2CO3 NH4Cl K2SO4