
SOLVED: Later this semester you will probably analyze a water sample from my well. Last year my well had a calcium level of 193 ppm. What would this concentration be if you

SOLVED: Calculating the Water Hardness For convenience assume that all hardness is due to the presence of only Caz+ions in the form of calcium carbonate, CaCO3 The unit of hardness may be
Solution Concentration Calculations (Molarity, molality, ppm, ppb) - SchoolTube - Safe video sharing and management for K12






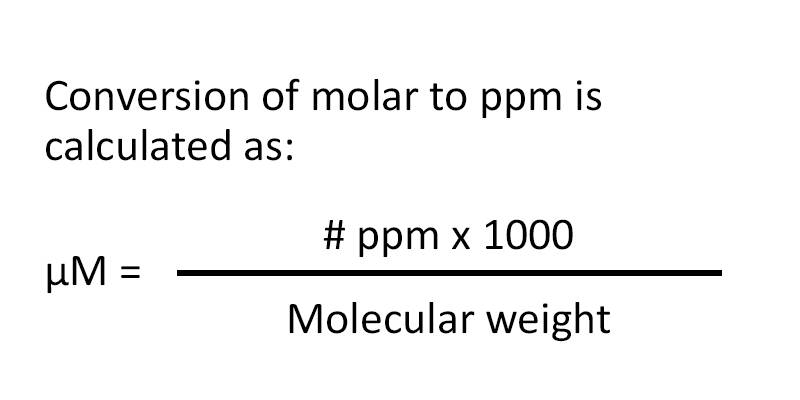
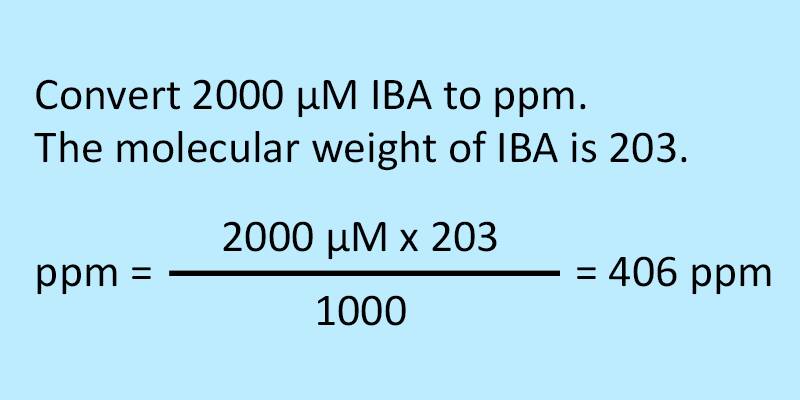




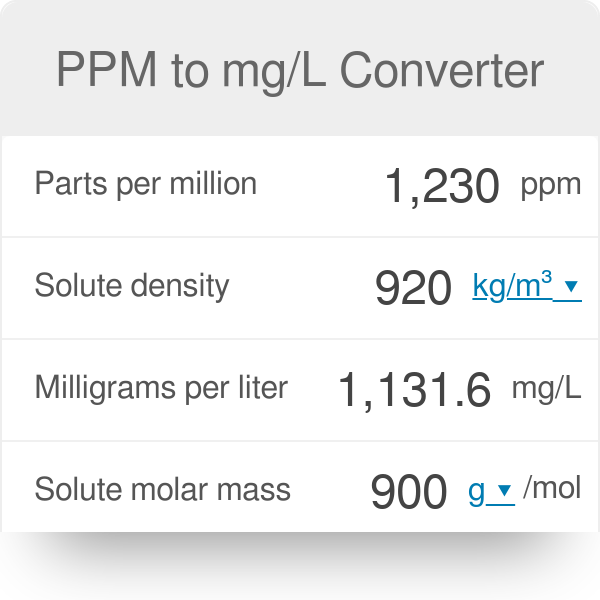
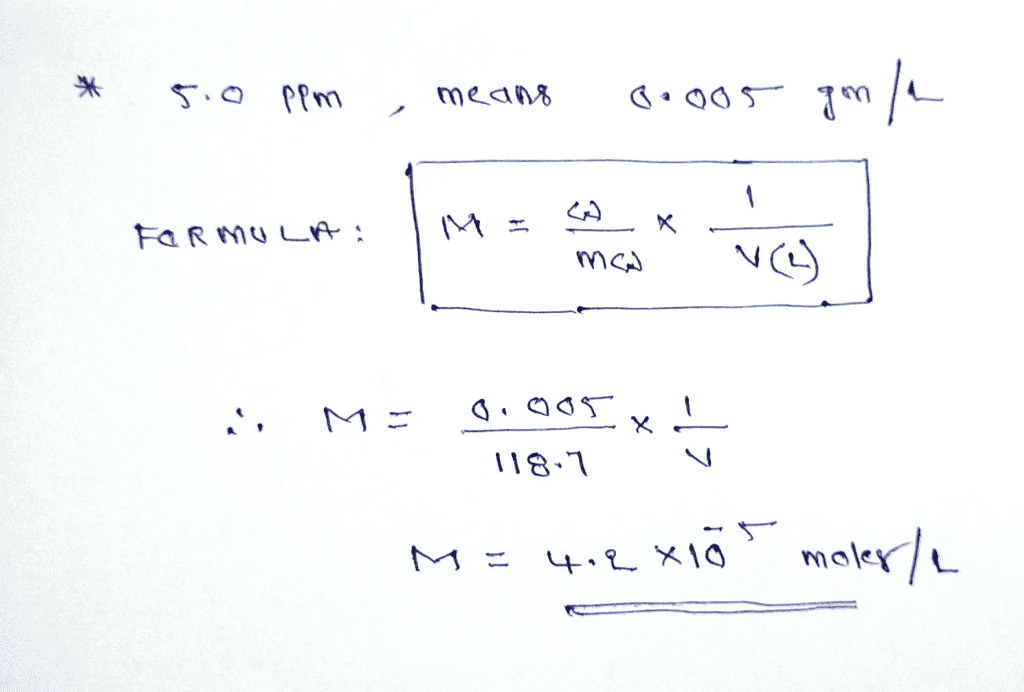
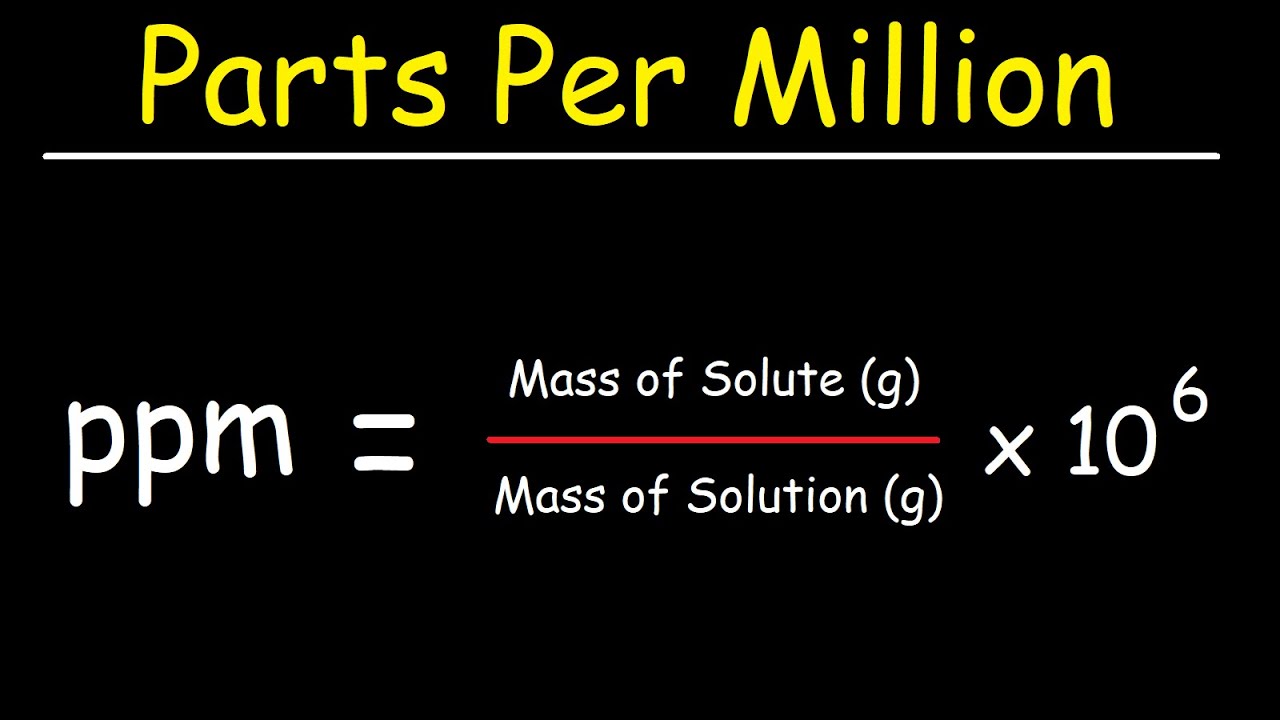
:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)


:max_bytes(150000):strip_icc()/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)
