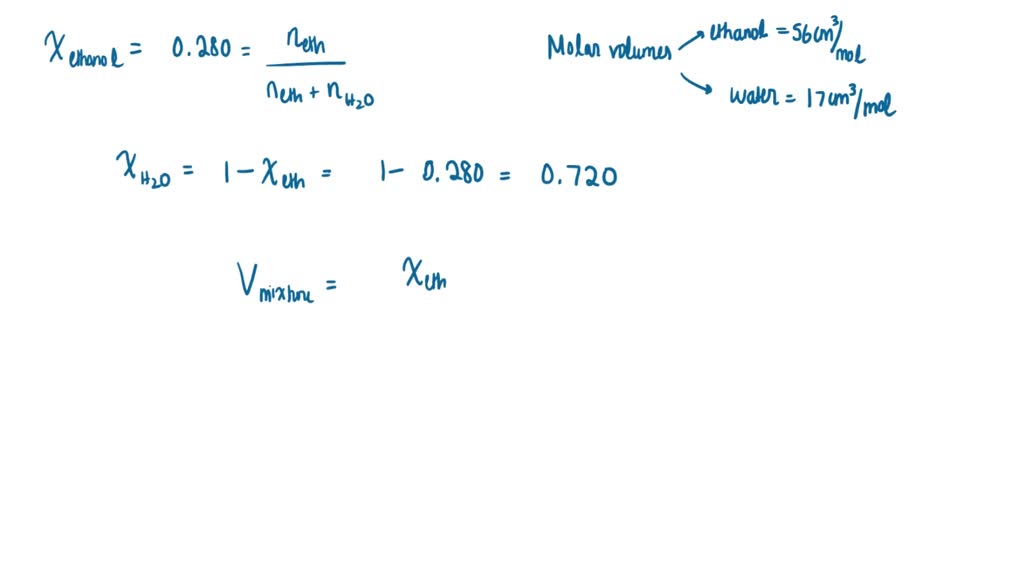
SOLVED: Calculate the volume of one mole (molar volume) of a mixture of water and ethanol in which mole fraction of ethanol is 0.280. Partial molar volumes of ethanol and water at

The molar volume of He at 10.1325 MPa and 273 K is 0.11075 of its molar volume at 101.325 kPa at 273 K. Calculate the radius of helium atom. The gas is

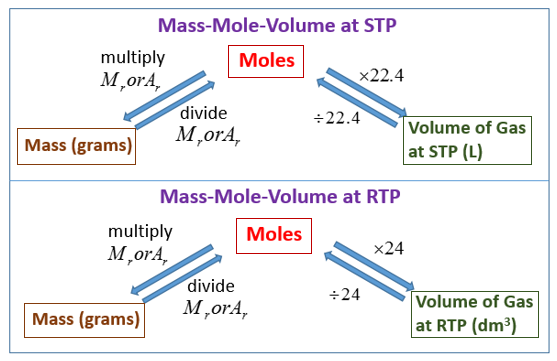
SOLVED:Use molar volume to calculate each of the following at STP: a. the number of grams of Ne contained in 11.2 L of Ne gas b. the volume, in liters, occupied by

SOLVED: What is the molar volume of ethane at 350 K and 70 bar according to: (a) the ideal gas law (b) the Van der Waal equation (a-5.562 Lzmol-Ibar) and (b= 0.0638

SOLVED:Use molar volume to calculate each of the following at STP: a. the number of moles of O2 in 44.8 L of O2 gas b. the volume, in liters, occupied by 0.420

Molar Volume Avogadro's Law: Equal volumes of gas at the same temperature and pressure contain equal numbers of particles. But the mass is different!!! - ppt download

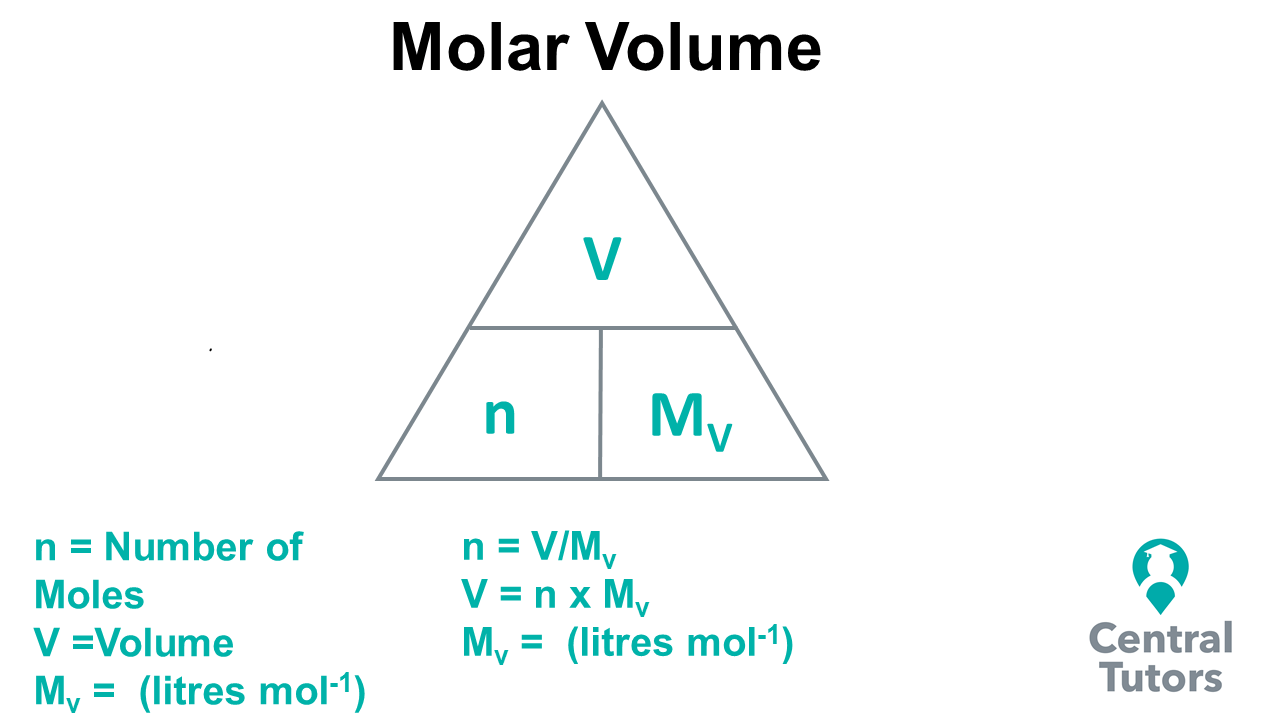
Mole and gas volume The molar volume of a gas is its volume per mole, litre mol-1. It is the same for all gases at the same temperature and pressure. The. -

Calculate standard molar volume of He ,if measurd density of He at stp is 0 1784g/l sir please solve - Chemistry - - 16212787 | Meritnation.com

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

Mole and gas volume The molar volume of a gas is its volume per mole, litre mol-1. It is the same for all gases at the same temperature and pressure. The. -