
Calculate the mass percent of different elements present in sodium sulphate ($N{{a}_{2}}S{{O}_{4}}$) - CBSE Class 11 Chemistry - Learn CBSE Forum

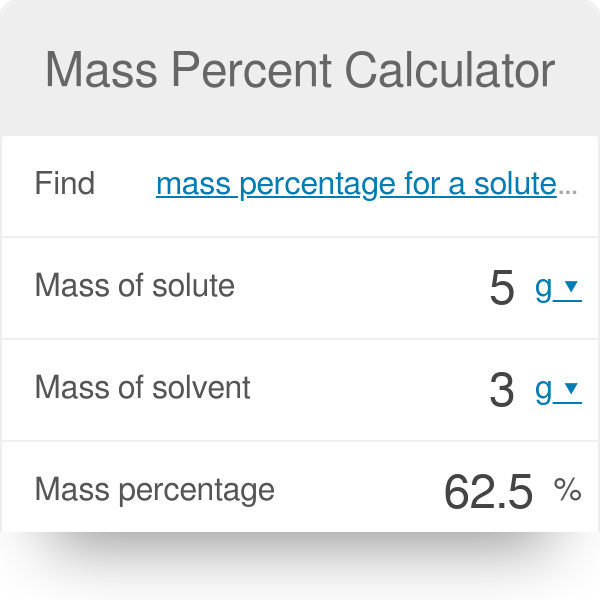
Mass Percent of a Solution Made Easy: How to Calculate Mass % or Make a Specific Concentration - YouTube

Question Video: Calculating the Percent by Mass of the Active Ingredient in an Anti-inflammatory Ointment | Nagwa

Calculate the mass percentage of carbon, hydrogen and oxygen in ethanol - CBSE Class 11 Chemistry - Learn CBSE Forum

SOLVED: Calculate the mass percent composition of carbon in each carbon-containing compound. a. CH4 b. C2H6 c. C2H2 d. C2H5Cl

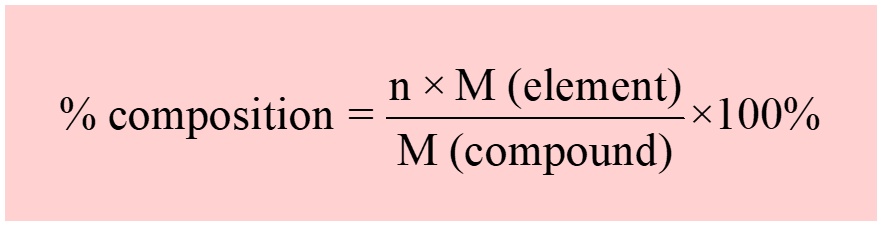


:max_bytes(150000):strip_icc()/Potassium-ferricyanide-58a279693df78c475811ba19.jpg)




:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)





